The leptospiremic (acute) phase and the immune (delayed) phase. You may have mild symptoms or no symptoms in the leptospiremic phase. Some people develop severe symptoms in the immune phase.
Leptospiremic phase
During the leptospirosis phase (also called the septicemic phase) you may experience a sudden onset of flu-like symptoms. This usually starts within two to 14 days after a Leptospira infection. It lasts between three and 10 days. In this phase, bacteria are in your bloodstream and moving to your organs. Blood tests will show signs of infection.
Immune phase
In the immune phase, Leptospira bacteria have moved from your blood to your organs. The bacteria are most concentrated in your kidneys, which make pee (urine). Urine tests will show signs of the bacteria and you’ll have antibodies to Leptospira in your blood. A small number of people will get very sick with Weil’s syndrome in this phase. Weil’s syndrome causes internal bleeding, kidney damage and severe yellowing of your skin and eyes (jaundice). In developing countries, the disease occurs most commonly in pest control, farmers, and low-income people who live in areas with poor sanitation. In developed countries, it occurs during heavy downpours and is a risk to pest controllers, sewage workers and those involved in outdoor activities in warm and wet areas. Diagnosis is typically by testing for antibodies against the bacteria or finding bacterial DNA in the blood. An estimated one million severe cases of leptospirosis in humans occur every year, causing about 58,900 deaths. The disease is most common in tropical areas of the world, but may occur anywhere. Outbreaks may arise after heavy rainfall. The disease was first described by physician Adolf Weil in 1886 in Germany. Infected animals may have no, mild, or severe symptoms. These may vary by the type of animal.
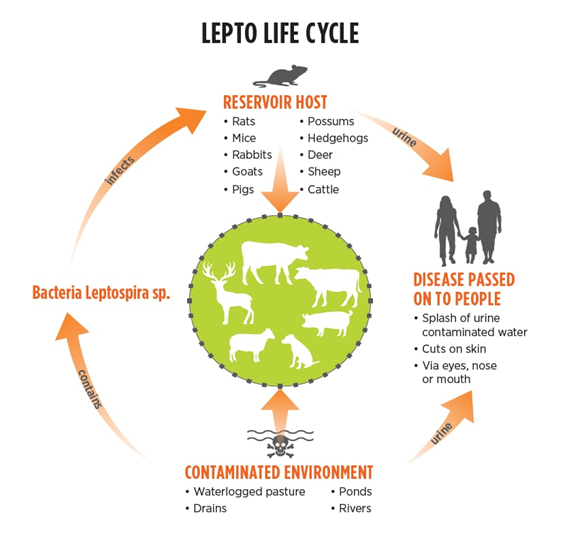
Life cycle of leptospirosis:
The life cycle of Leptospira bacteria involves an enzootic cycle in animal hosts and survival in the environment, with humans being incidental or accidental hosts. The cycle has two main stages: persistence in a reservoir host and environmental transmission.
1. Persistence in a Reservoir Host
Many wild and domestic mammals serve as natural, long-term reservoirs for Leptospira bacteria, usually without showing severe symptoms.
Colonization: After initial infection, the bacteria travel through the bloodstream and colonize the renal tubules of the kidneys.
Shedding: The infected animals (such as rodents, dogs, cattle, and pigs) continuously or intermittently excrete large amounts of viable bacteria in their urine into the environment for extended periods, sometimes years or their entire lives. This is the key mechanism by which the bacteria are maintained in nature.
2. Environmental Transmission
The Leptospira bacteria are able to survive for weeks to months in favourable environmental conditions, outside a host.
Survival conditions: The bacteria thrive in warm, moist, and stagnant freshwater or damp soil with a neutral to slightly alkaline PH. They are susceptible to drying, freezing, and high temperatures.
Transmission to new hosts: A new animal or human host becomes infected through direct contact with infected animal urine or indirectly through contact with a contaminated environment (water, soil, mud). The bacteria enter the body through cuts, abrasions, or intact mucous membranes (eyes, nose, mouth). Ingestion of contaminated water or food is also a possible route of infection.
3. Human Infection
Humans are considered accidental, dead-end hosts in the leptospirosis life cycle.
Biphasic illness: In humans, the infection typically manifests as a biphasic illness. The initial acute/leptospiremic phase involves the bacteria in the bloodstream and rapid dissemination to organs like the liver, kidneys, and lungs. The immune phase follows, where antibodies clear the bacteria from the blood but organ damage (e.g., kidney damage, liver failure, pulmonary haemorrhage) can occur due to the immune response.
Limited shedding: While humans can shed the bacteria in their urine during the immune phase, this is typically for a much shorter duration (weeks to a couple of months) compared to reservoir animals, and person-to-person transmission is extremely rare.
Humans are accidental hosts, infected incidentally after animal or environmental exposure. Transmission of the organism to humans occurs via portals of entry, including cuts or abraded skin, mucous membranes, or conjunctivae Human exposures that lead to infection include contact with urine-contaminated soil or water (eg, floodwater, ponds, rivers, streams, sewage), ingestion of food or water contaminated by urine or urine-contaminated water, or direct contact with the urine or reproductive fluids from infected animals [10]. Transmission has also rarely occurred through animal bites. Controversy exists as to whether Leptospira can penetrate intact skin. Human-to-human transmission is very rare but has been documented through sexual intercourse and breastfeeding reproductive tract, leading to transmission during mating.
Pathophysiology:
Leptospirosis pathophysiology involves bacteria entering the body through broken skin or mucous membranes, spreading via the bloodstream to organs like the liver and kidneys, and causing damage through direct bacterial action and a strong immune response. In severe cases (Weil's syndrome), this can lead to a life-threatening cascade of organ damage including jaundice, acute kidney injury (AKI), pulmonary hemorrhage, and a systemic inflammatory response that can include a cytokine storm followed by immunoparalysis.
Entry and spread
Entry: Leptospira bacteria enter the body, often through nonintact skin or mucous membranes, from contact with infected animal urine, contaminated water, or soil.
Spread: After entry, the bacteria travel through the lymphatic system before entering the bloodstream (bacteremia), from which they spread throughout the body.
Seeding: The bacteria colonize various organs, particularly the liver and kidneys, where they multiply.
Mechanisms of damage
Bacterial damage: The bacteria can cause direct damage to cells and tissues, including the endothelial lining of capillaries, which impairs blood flow. A protein called hemolysin SphH can form pores in cell membranes, leading to cell damage and hemorrhages.
Immune-mediated damage: The body's immune response is a major driver of tissue and organ damage.
Inflammatory response: The initial immune response involves cytokine production.
Cytokine storm and immune paralysis: In severe cases, an excessive and uncontrolled release of cytokines can lead to a "cytokine storm," followed by a state of immune paralysis, which is a feature of sepsis and can lead to organ failure.
Organ-specific effects
Liver: Direct damage to hepatocytes disrupts intercellular junctions, leading to liver dysfunction, jaundice, and elevated liver enzymes.
Kidneys: Bacteria colonize the renal tubules. This, along with the immune response, leads to renal damage and acute kidney injury (AKI). Renal failure is the most common cause of death.
Lungs: The condition can cause pulmonary hemorrhage, a leading cause of mortality in severe cases.
Other organs: The infection can manifest in various other ways, including nervous system damage, ocular problems, and hematological abnormalities.
Data/Statistics and History:
Leptospirosis occurs worldwide but is endemic mainly in countries with humid subtropical and tropical climates. Estimates indicate that there are more than 500,000 cases of leptospirosis each year worldwide. Leptospirosis is a disease of epidemic potential, especially after heavy rainfall or flooding. Cases have been reported in most countries of the Americas and outbreaks have been reported in Brazil, Nicaragua, Guyana and several other Latin American countries. The majority of reported cases have severe manifestations, for which mortality is greater than 10%. The number of human cases is not known precisely due to under- or misdiagnosis. Outbreaks can be associated with floods and hurricanes. Leptospirosis can also be an occupational hazard for people who work outdoors or with animals, such as rice and sugar-cane field workers, farmers, sewer workers, veterinarians, dairy workers, and military personnel. It is also a recreational hazard to those who swim or wade in contaminated water. Leptospirosis is a problem of human and veterinary public health. The numerous Leptospira strains can establish infections within a variety of animal hosts that includes rodents, livestock, and other domestic animals while humans serve as incidental hosts. Wild and domestic animals in the carrier state may shed leptospires intermittently for many years or even a lifetime.
History:
Leptospirosis has a long history, recognized in ancient texts as "rice field jaundice" and other names before physician Adolf Weil formally described its severe form, Weil's disease, in 1886. The causative agent, a spiral-shaped bacterium, was discovered in 1907 by Stimson, and later identified independently in Japan and Germany in 1915. The role of rats as a vector was discovered in 1917.
Early observations and recognition
Ancient times: Diseases matching leptospirosis were described in ancient Chinese and Japanese texts as occupational hazards, particularly for rice field workers.
1812: The French physician Larrey described a similar disease, "fièvre jaune," among Napoleon's troops in Cairo.
1886: Adolf Weil published his description of the severe form of the disease, which became known as Weil's disease, noting the symptoms of jaundice, fever, hemorrhage, and kidney failure.
1907: The spiral-shaped bacterium was first observed in the kidneys of a patient with Weil's disease by Stimson, who named it Spirochaeta interrogans.
1915-1916: Scientists in both Japan and Germany independently demonstrated that the Spirochaeta interrogans was the cause of the disease.
Later discoveries and control
1917: The role of rats as a source of human infection was discovered.
1940s: The disease became a known occupational hazard for sewer workers and others exposed to contaminated water.
Mid-1900s: Researchers developed vaccines to help prevent the disease.
Today: Leptospirosis is recognized as a global zoonotic disease and a significant public health concern, particularly in tropical regions.
In ancient China, a disease that was certainly leptospirosis was recognized as an occupational hazard of rice harvesters. In Japan, leptospirosis was called akiyami, or autumn fever, a term still used for this disease. In the West, leptospirosis was described by Larrey in 1812 as fièvre jaune among Napoleon's troops at the siege of Cairo. The disease initially was believed to be related to the plague but not as contagious. Throughout the remainder of the 19th century, leptospirosis was known in Europe as bilious typhoid. Leptospirosis was recognized as an occupational disease of sewer workers in 1883. In 1886, Adolph Weil published his historic paper describing the most severe form of leptospirosis that would be later known as Weil disease. Weil described the clinical manifestations in 4 men who had severe jaundice, fever, and hemorrhage with renal involvement. In 1907, Stimson used special staining techniques in the postmortem examination of a kidney from a person with Weil disease and found a spiral organism with hooked ends, which was named Spirochaeta interrogans because its shape resembled that of a question mark. Inada et al identified the causal agent of infectious jaundice in Japan in 1916, naming the organism Spirochaeta icterohaemorrhagiae.
Case Description:
We report a case of leptospirosis with tick-borne typhus coinfection in an abattoir worker who presented with a short history of fever, myalgia, jaundice, nonoliguric renal failure, diffuse petechial rash, and altered sensorium. His lab investigations showed leukocytosis, raised C-reactive protein (CRP), elevated transaminases and creatinine, mild pleocytosis, and mildly raised proteins in cerebrospinal fluid (CSF). Serology for Leptospira IgM was positive by enzyme-linked immune sorbent assay (ELISA). A paired Weil-Felix test (WFT) showed a fourfold increase in OX19 and OX2 titers. The patient responded well to IV antibiotic therapy and was discharged. This is the first time that leptospirosis and Indian tick-borne typhus coinfection has been reported from western India.
Taxonomy and classification:
Leptospirosis is classified in two main ways: by genotype (based on DNA) and by serovar (based on antigenic properties). Genetically, the genus Leptospira is divided into species, with some being pathogenic and others non-pathogenic. Antigenically, a more traditional classification groups serovars into serogroups, though this doesn't align perfectly with genetic classifications. For example, Leptospira interrogans was once the species for all pathogenic strains, but modern classification uses more specific species based on DNA.
Classification by genotype
Genus: Leptospira
Species: There are 21 species, with 9 being pathogenic and 5 intermediates. The pathogenic strains are typically classified under L. interrogans.
Clades/Subclades: Based on genomic sequences, Leptospira are further divided into two clades and four subclades (P1, P2, S1, and S2).
The P1 subcase contains species that can cause severe disease.
Classification by serovar and serogroup
Serovar: Defined by serological (antigenic) testing, not DNA. There are around 240 identified serovars, of which only a few are pathogenic.
Serogroup: A grouping of serovars that are antigenically related.
Serogroups have been useful for epidemiological purposes but do not have official taxonomic standing, as genetically different strains can sometimes fall into the same serogroup.
Nomenclature: The accepted taxonomic format is Genus species serovar name (e.g., Leptospira interrogans serovar Ballum).
Traditional vs. modern classification
Historically: Leptospira interrogans was used for all pathogenic strains, and L. biflexa for all saprophytic strains.
Modern: Genomic and genetic data have led to the current, more complex classification system, which acknowledges more species and better distinguishes between pathogenic and non-pathogenic strains.
Animal and environmental reservoirs:
Mammals act as the primary reservoir for Leptospira organisms. The environment can serve as a reservoir if it becomes contaminated by the urine of infected mammals.
- Animal reservoir – Approximately 160 mammalian species have been identified as natural carriers of pathogenic Leptospira species [11]. The organism lives in the renal tubules of infected mammals and is shed in the urine.
Rodents are the most important reservoirs for maintaining transmission in most settings. Infection in small rodents (eg, rats) usually occurs in utero, during birth, or during infancy from environmental contamination of the nest [12]. Once infected, rodents are asymptomatic carriers and shed the organism in their urine intermittently or continuously throughout life, resulting in contamination of the environment, particularly water In addition to rodents, the organism infects a variety of both wild and domestic mammals, especially cattle, swine, dogs, horses, sheep, and goats. It rarely occurs in cats. Animals can be asymptomatic carriers or can develop clinical infection, which may be fatal. Mortality in dogs is estimated at approximately 10 percent. Spontaneous abortion is a common outcome of leptospirosis in cattle, swine, sheep, and goats.
- Environmental reservoir – Organisms can survive for days to months in urine-contaminated soil and fresh water [10].
Contamination of water with animal urine can occur via several mechanisms:
• Following heavy rainfall or flooding that allows mixing of urine-contaminated soil or sewage with water, especially in areas with poor housing and sanitation/sewage conditions that propagate high rodent population’s Animal excretion of urine into wet soil or bodies of fresh water, such as ponds, lakes, rivers, and streams
• Drainage of urine-contaminated soil or water into bodies of fresh water
Causes:


 Kumar Raja Jayavarapu*
Kumar Raja Jayavarapu*
 K. Uma Maheswari
K. Uma Maheswari


 10.5281/zenodo.17694825
10.5281/zenodo.17694825